Properties of Water Notes
Water is made up of two parts hydrogen and one part oxygen. Each hydrogen atom shares its only electron with the oxygen, and the oxygen atom shares one of its electrons with the hydrogen atom. These shared electrons stay near the oxygen atom more than the hydrogen atoms. The hydrogen atoms carry a slightly positive charge, whereas the oxygen atoms carry a slightly negative charge.
Hydrogen Bonding- The weak attraction between the positive hydrogen atoms and negative oxygen atoms.
Hydrogen Bonding- The weak attraction between the positive hydrogen atoms and negative oxygen atoms.
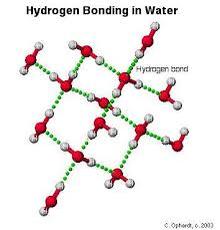 An example of hydrogen bonding.
An example of hydrogen bonding.
This is what happens in hydrogen bonding.
Water is a universal solvent, meaning it can dissolve anything. Substances "afraid" of water are called hydrophobics.
Cohesion- When water molecules attract to each other.
Cohesion- When water molecules attract to each other.
Adhesion- When water molecules attract to each other. You can see an example of adhesion when your clothes get wet, as the water sticks on your clothes.
Surface Tension- When water molecules attract to create a "film". Surface tension is responsible for small bugs being able to walk across water without them falling in. Surface tension also helps small things like paperclips to stay at the surface. However, the paperclip is unable to stay afloat the surface, as the weak bonds most likely break.
Temperature- Measure of the average kinetic energy of the atoms and molecules measured in degrees (Celsius)
Heat- Measure of the total kinetic energy of the atoms and molecules. It is measured in Calories.
Calories- The amount of heat needed to raise the temperature of 1 gram of water by 1 Celsius.
Heat- Measure of the total kinetic energy of the atoms and molecules. It is measured in Calories.
Calories- The amount of heat needed to raise the temperature of 1 gram of water by 1 Celsius.
High Heat of Vaporization- Hydrogen bonds break so water can absorb heat without changing phase. This allows your sweat to help you cool off after exercising.
High Latent Heat of Fusion- Heat is released when water freezes and heat is absorbed when ice melts.
Water temperature is more stable than air temperature and water moderates climates. Some of the breezes we feel can come from the ocean. Water has a high specific heat, as it can absorb a large amount of heat before it begins to get hot.
Density is how much stuff you can compact in a space. To find density, you would do mass/volume. Interestingly, water is less dense in its solid form.
Density is how much stuff you can compact in a space. To find density, you would do mass/volume. Interestingly, water is less dense in its solid form.
KHS- Marine Science: Investigating Water Lab
Evaporative Cooling: Station #1 Water and Alcohol

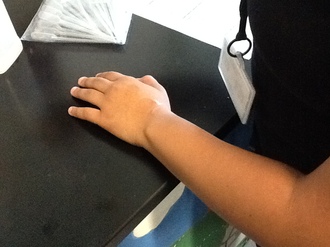
In this station, we had to test which felt cooler on our hand; water or alcohol. We placed a drop of water and alcohol at the back of our hands with the droppers in the picture to the left. Our group felt the alcohol was cooler than the water. However, the alcohol would not cool us down.
Why is it beneficial that your sweat is water and not alcohol based?
It is beneficial to sweat water than alcohol because alcohol dries faster, leaving you to be too hot, as the alcohol isn't taking away the excess heat like water would.
Why is it beneficial that your sweat is water and not alcohol based?
It is beneficial to sweat water than alcohol because alcohol dries faster, leaving you to be too hot, as the alcohol isn't taking away the excess heat like water would.
Water Tension/Cohesion - Station #2 Water on a Penny

Before we started this station, we had to predict how many water drops would fit on a penny (I guessed 13). When the experiment began, we used the pipette to add water one drop at a time until the penny overflowed with water. As we kept adding drops, more and more water would build up on top of the penny. Our penny overflowed at 24 drops.
How many drops fit on the penny? How was your penny able to hold that many drops?
Our penny was able to hold 24 drops of water. The penny was able to hold many drops due to the cohesion (and a bit of adhesion) of the water. Since the water molecules were able to stick together, they were able to build on top of the penny until they couldn't hold anymore.
How many drops fit on the penny? How was your penny able to hold that many drops?
Our penny was able to hold 24 drops of water. The penny was able to hold many drops due to the cohesion (and a bit of adhesion) of the water. Since the water molecules were able to stick together, they were able to build on top of the penny until they couldn't hold anymore.
Surface Tension: Station #3- Water on Parafilm

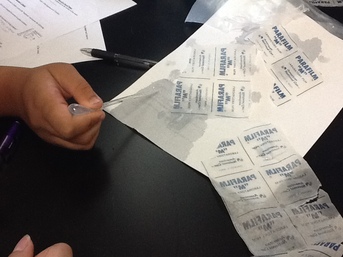
What we did with this station was drop water with the pipettes on Parafilm and regular paper. As we watched the papers absorb the water, our group noticed the Parafilm would absorb the water faster than the regular paper.
Surface Tension Station #4- Paper Clips on the Surface of a Cup of Water

Just like the penny lab, we estimated how many paper clips we could float on the surface of water. I originally put 6, but changed it to one, as this lab soon proved to be difficult to get even one paper clip floating. We used tweezers and non-shaky hands to carefully place a paper clip on the surface of the water. However, our dreams were crushed, since the paper clip would sink over and over again. However, we were able to get one paper clip to float, nearly causing everyone around Station 4 to start a party.
Carefully add one drop of dish-soap to the water- what happens now?
If we were to add one drop of dish-soap to the water, the paper clip (if any) floating on the surface of the water would sink. This is because the hydrogen bonds would break, causing the paper clip to sink.
Carefully add one drop of dish-soap to the water- what happens now?
If we were to add one drop of dish-soap to the water, the paper clip (if any) floating on the surface of the water would sink. This is because the hydrogen bonds would break, causing the paper clip to sink.
Adhesion: Station #5- Graduated Cylinder

In this station, we looked at water in a graduated cylinder. We observed the meniscus (the curved surface of liquid in a tube) to measure how much water was in the graduated cylinder. The water line in the graduated cylinder is not a straight line because of the adhesion the water is doing.
Why would the water property of adhesion be important in living systems?
The water property of adhesion is important in living systems because it helps cool us down, and some living systems need water travelling in them.
Why would the water property of adhesion be important in living systems?
The water property of adhesion is important in living systems because it helps cool us down, and some living systems need water travelling in them.
Capillary Action- Station #6- Rolled up Paper Towel

In this station, we had two 250 mL beakers, blue food coloring, and a paper towel. We filled up one of the beakers with water and put blue food coloring in it. Then we rolled the paper towel and placed it in the two beakers. This is to show how water transfers from one area to the other. Eventually, there would be the same amount of water in each beaker.
Why would capillary action be important to plants and trees? Explain.
Capillary action is important to plants and trees because they need water to survive. The capillary action would help move water in plants so they can stay healthy and alive.
Why would capillary action be important to plants and trees? Explain.
Capillary action is important to plants and trees because they need water to survive. The capillary action would help move water in plants so they can stay healthy and alive.